Hoofddorp, Netherlands, 5 August 2020
This phase 2/3 multi-center, randomized, double-blind trial investigated the effect of a new 3.06% diclofenac sodium gel formulation (AMZ001) for the relief of knee OA pain, in 444 patients exposed to either AMZ001 once daily (QD), AMZ001 twice daily (BID) or placebo. The trial also included an exploratory comparator treatment arm, Voltaren® Gel 1 % four times daily (QID)[i].
For the primary analysis of change from baseline to week 4 in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain sub-score, AMZ001 QD showed a nominally significant improvement in reducing the pain compared to placebo (p<0.05). The results suggest that differences for BID were borderline significant (p<0.10).
The post-hoc analysis, including patients meeting WOMAC pain inclusion criterion at screening and baseline (89% of enrolled subjects), demonstrated nominally statistically significant reductions in WOMAC pain sub-scores compared to placebo for AMZ001 BID (p=0.029) and AMZ001 QD (p=0.019); differentiation to placebo was also strengthened for efficacy endpoints, as shown in Figure 1b.
While the study design and differences in samples sizes prevent formal comparisons between the AMZ001 treatment arms and the active comparator arm, there were no statistically significant differences between Voltaren® gel 1% QID compared with placebo or any AMZ001 regimens for all endpoints.
The safety and tolerability of AMZ001 was favorable with a low frequency of adverse events leading to discontinuation (2.5% to 6.6%) comparable to Voltaren® Gel 1% (6.2%). No serious adverse events were reported during the trial.
Dr. Lee S. Simon, Rheumatologist commented: “It is clear that topical Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) can be important therapies for patients with osteoarthritis of the knee. Guidelines recommend these therapies as first line in patients who do not respond to weight loss and exercise alone.
The results of this study are very encouraging for the hundreds of millions of patients with knee OA which represent 80% of the total burden of OA. Reducing the number of applications per day could help with adherence and thus help to reduce the chronic pain that could be caused by this disease.”
The study also found regarding secondary endpoints that the patients treated with AMZ001 experienced consistent improvements in quality of life (EQ-5D VAS questionnaire), in patient global assessment (PGA) and in productivity and activity (Work Productivity and Activity Index - WPAI) as shown in Figure 1a. These results are encouraging, considering that arthritis/rheumatism is the third leading cause of work disability in the Unites States after back/neck problems and depression/anxiety[ii]. Systemic exposure data indicated that AMZ001 delivers the active agent to the targeted tissue reducing the exposure to other body systems. This local action could be an asset in an elderly patient population who is usually on several drugs for ageing-associated diseases.
Dario N. Carrara PhD, Chief Scientific Officer commented: “This is the first study that investigated the efficacy of topical diclofenac on a once daily regimen in patients suffering from osteoarthritis of the knee. Our product takes advantage of the company’s proprietary topical transdermal technology to decrease the need for treatment to once a day in comparison to Voltaren® 1% four times daily gel.”
Fig. 1. Statistical analysis of treatment differences (95% CI) between placebo vs AMZ001 BID, AMZ001 QD, and Voltaren® 1% for the a) planned and b) post-hoc analysis of primary and key secondary endpoints (mITT set)[iii]
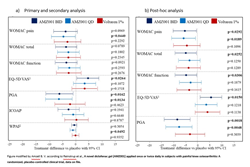
1 Positive value indicate improvement.
2 WPAI scores presented for overall work impairment sub-score, normalized 0–100.
Note: WOMAC, EQ-5D VAS, PGA and ICOAP scores are normalized 0–100.
BID: twice daily; CI: confidence interval; EQ-5D: EuroQol-5 Domain Questionnaire; ICOAP: intermittent and constant OA pain; mITT: modified intention-to-treat; OA: osteoarthritis; PGA: Patient Global Assessment;
QD: once daily; VAS: visual analog scale; WOMAC: Western Ontario and McMasters Universities Osteoarthritis Index; WPAI: Work Productivity and Activity Impairment questionnaire.
Note: Treatment differences vs placebo (95% CI) are presented for all endpoints; scores were normalized 0–100.
About Amzell
Amzell B.V is a specialist, virtual development pharmaceutical company which takes candidate or repurposed drugs and devices either through to proof of concept, or through to registration for sale to commercial healthcare companies.
Amzell B.V has particular interest in developing well-characterized active substances utilizing innovative platform drug delivery technologies, with the aim to provide more effective drug delivery, increased efficacy, and improved safety and compliance.
AMZ001 - topical diclofenac gel for the treatment of pain related to knee osteoarthritis
AMZ001 is a non-invasive topical gel formulation containing 3.06% diclofenac sodium which is a nonsteroidal anti-inflammatory drug (NSAID). Each dose of AMZ001 is accurately administered by an airless metered dose pump.
Its higher drug strength (3.06%) combined with an efficient topical delivery system allows AMZ001 to improve localized pain with a single daily application (QD), with a lower total dose compared to other topical NSAIDs treatments.
[i] www.clinicaltrials.gov; ClinicalTrials.gov Identifier: NCT03691844
[ii] Vina ER, Kwoh CK. Epidemiology of osteoarthritis: literature update. Curr Opin Rheumatol 2018;30:160–7.
[iii] Bihlet A.R, Byrjalsen I, Nielsen H.B, Andersen J.R, Derne C, Delpy L, Carrara D, A placebo-controlled, double-blind, randomized, trial of AMZ001 - a novel diclofenac sodium 3.06% gel - for the treatment of knee osteoarthritis symptoms. Abstract 725. Osteoarthritis and Cartilage, volume 28,supplement 1, S484, 01 April 2020.